As one of the top universities in China, the University of Science and Technology of China (USTC) has a strong research presence in the field of power cells. Now, they have seen a new result.
A team at the university has synthesized ultra-small platinum-based intermetallic compound electrocatalysts that allow proton exchange membrane fuel cells to maintain 81.5 percent of their discharge power after 30,000 cycles of endurance testing, according to information published on their website last week.
The data means that if it can be used in production, it is expected to allow such fuel cells to last tens of times longer than ordinary power cells.
Among the power batteries in common use today, cadmium-nickel batteries have a charge-discharge cycle life of 500 to 800 times, lead-acid batteries 200 to 500 times and lithium-ion batteries 600 to 1,000 times.
In an article earlier this month, Ouyang Minggao, a member of the Chinese Academy of Sciences, said that for private cars, a battery cycle life of 1,000 cycles would be sufficient.
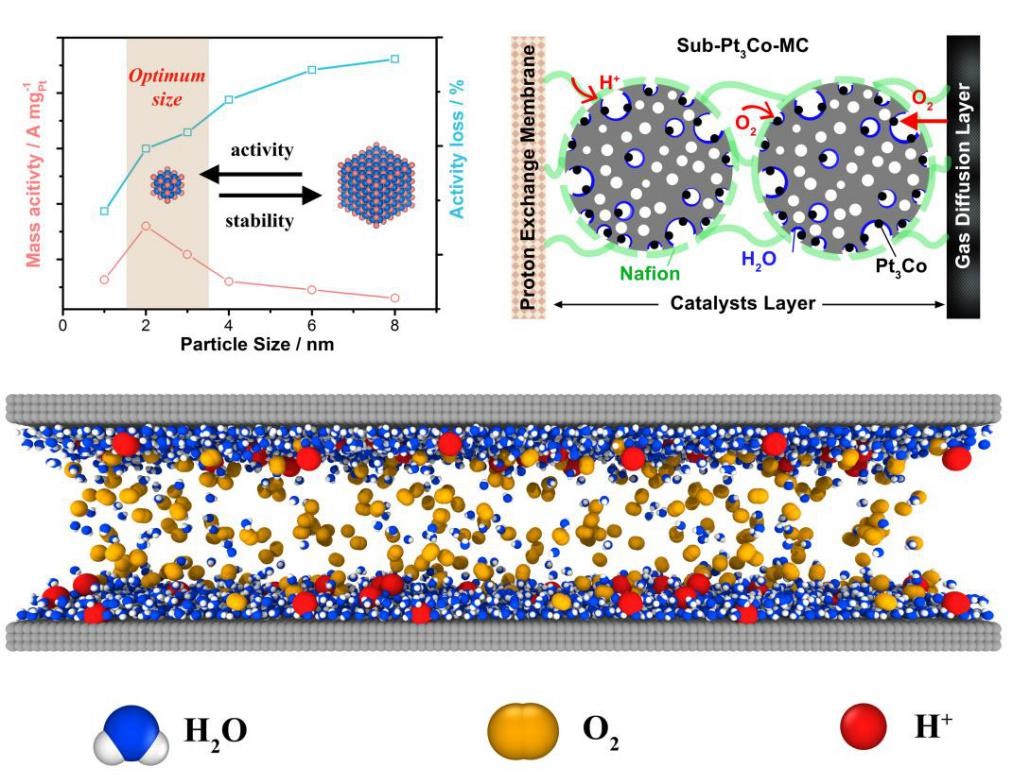
The research, conducted by the university's team of Professor Wu Changzheng of the School of Chemistry and Materials Science and Professor Wu Hengan of the Department of Modern Mechanics in the School of Engineering Sciences, revealed the mechanism of mass transfer at the three-phase interface within the battery.
Their results have been published in the Proceedings of the National Academy of Sciences (PNAS) at the following link.
https://www.pnas.org/content/118/35/e2104026118
Proton exchange membrane fuel cells have advantages over other types of cells such as high discharge power and no pollution, in which the cathode oxygen reduction reaction is the rapid control step of the full cell reaction.
Platinum-based intermetallic compounds have a natural advantage in stability and are the next generation of commercial oxygen reduction catalyst systems for fuel cells, according to the article published on the USTC website.
Currently, platinum-based intermetallic compounds still have problems such as large particle size, resulting in reduced platinum utilization and mass activity, which has become one of the key bottlenecks limiting fuel cell performance improvement, the article published said.
(Image source: the USTC website)
To address this challenge, Prof. Wu Changzheng's team synthesized Pt3Co, PtCo, and Pt3Ti particles with ultra-small Pt-based intermetallic compounds of about 2 nm in size.
The research optimizes the structural design and achieves high-performance expression of catalysts in fuel cell membrane electrodes, providing new ideas for catalytic materials for fuel cell cathodes, according to the article.
USTC's research strength is almost comparable to that of Tsinghua University and Peking University in China. The university is located in Hefei, Anhui province, which is also home to Nio China and the JAC Nio facility.
Last month, Professor Ma Cheng's team at the university announced that they had successfully developed a new chloride solid electrolyte material, lithium zirconium chloride.
Lithium zirconium chloride is the first high-performance chloride solid-state electrolyte based on tetravalent cations, overcoming the bottleneck of difficulty to combine production cost and comprehensive performance, and is expected to advance the commercialization of all-solid-state batteries, according to the team.
On April 30, Nio signed a strategic cooperation agreement with the USTC to cooperate in technology research and talent training.
On August 4, Nio and the USTC jointly established the Smart Electric Vehicle Joint Laboratory to officially launch.
The lab announced the development plan and project operation mechanism for 2021-2022, and released more than twenty intended topics including autonomous driving system engineering, autonomous driving algorithm, intelligent hardware, information security and big data, electric core material, and power management.
According to the plan, the joint lab will select a number of projects with cutting-edge topics to enter the joint R&D stage in September this year.



